
This is actually one of the chemical properties of metals and nonmetals: metals tend to form cations, while nonmetals tend to form anions. First, each element that forms cations is a metal, except for one (hydrogen), while each element that forms anions is a nonmetal. There are several things to notice about the ions in Table 3.6 “Monatomic Ions of Various Charges”. Table 3.6 “Monatomic Ions of Various Charges” lists some common ions in terms of how many electrons they lose (making cations) or gain (making anions). When atoms gain or lose electrons, they usually gain or lose a characteristic number of electrons and so take on a characteristic overall charge. When they do, they become monatomic ions. Individual atoms can gain or lose electrons. Compounds formed from positive and negative ions are called ionic compounds. Remember that ions are formed only when electrons move from one atom to another a proton never moves from one atom to another. Species with overall positive charges are termed cations, while species with overall negative charges are called anions. Electrons can move from one atom to another when they do, species with overall electric charges are formed. They have the same number of electrons as protons, so the negative charges of the electrons is balanced by the positive charges of the protons. So far, we have discussed elements and compounds that are electrically neutral. 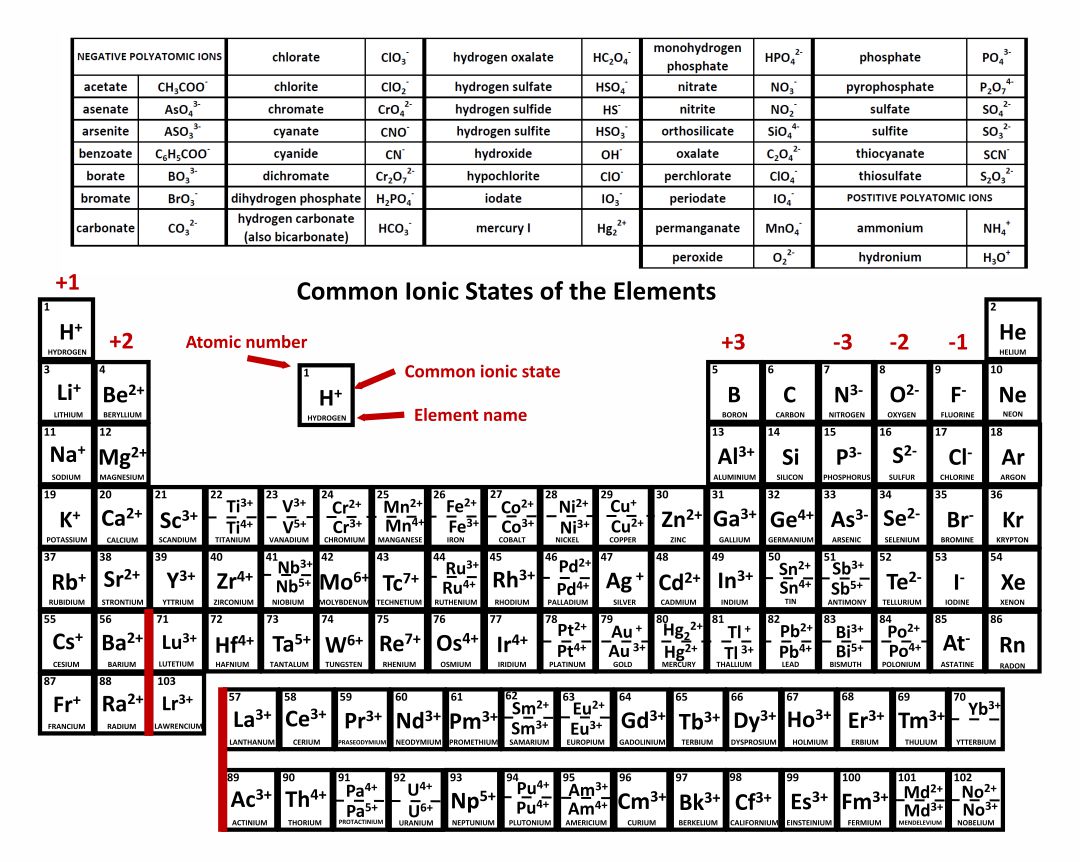
Generate a proper name for an ionic compound.Construct a proper formula for an ionic compound.Learn the characteristic charges that ions have.Read about electron configurations to learn more. This gives them a full S subshell, which is a pretty stable configuration. For example, metals often lose enough electrons to leave two valence electrons remaining (example: carbon family with +2 charge). Most elements have the ability to form more than one charge, but there are patterns to be found there, as well.You can check out this fancier Periodic Table to see the most common charges of most of the elements.Therefore, sometimes that row is thought of as having “+/- 4” charge, which fits nicely between the +3 and -3 columns. But it’s possible for carbon and silicon to form a C 4- / Si 4- ions, and all of that family’s elements can form +4 ions. The elements in the carbon family aren’t listed as +4 or -4 charged ions, because they are more likely to form covalent bonds instead of being ions.For example, iron can form Fe 2+ and Fe 3+ ions, depending on the situation. The transition metal elements (middle section of the table) don’t have the same type of predictable charge patterns, and most of them are capable of forming more than one type of ion.But at least we can say that it’s capable of forming a P 3- ion to fit the pattern shown in the chart. For example, phosphorus is actually more likely to be found with a positive charge in a compound with oxygen. These elements don’t always follow such this pattern.(The numbers at the top of the columns show what charge that family’s elements sometimes have as ions.) 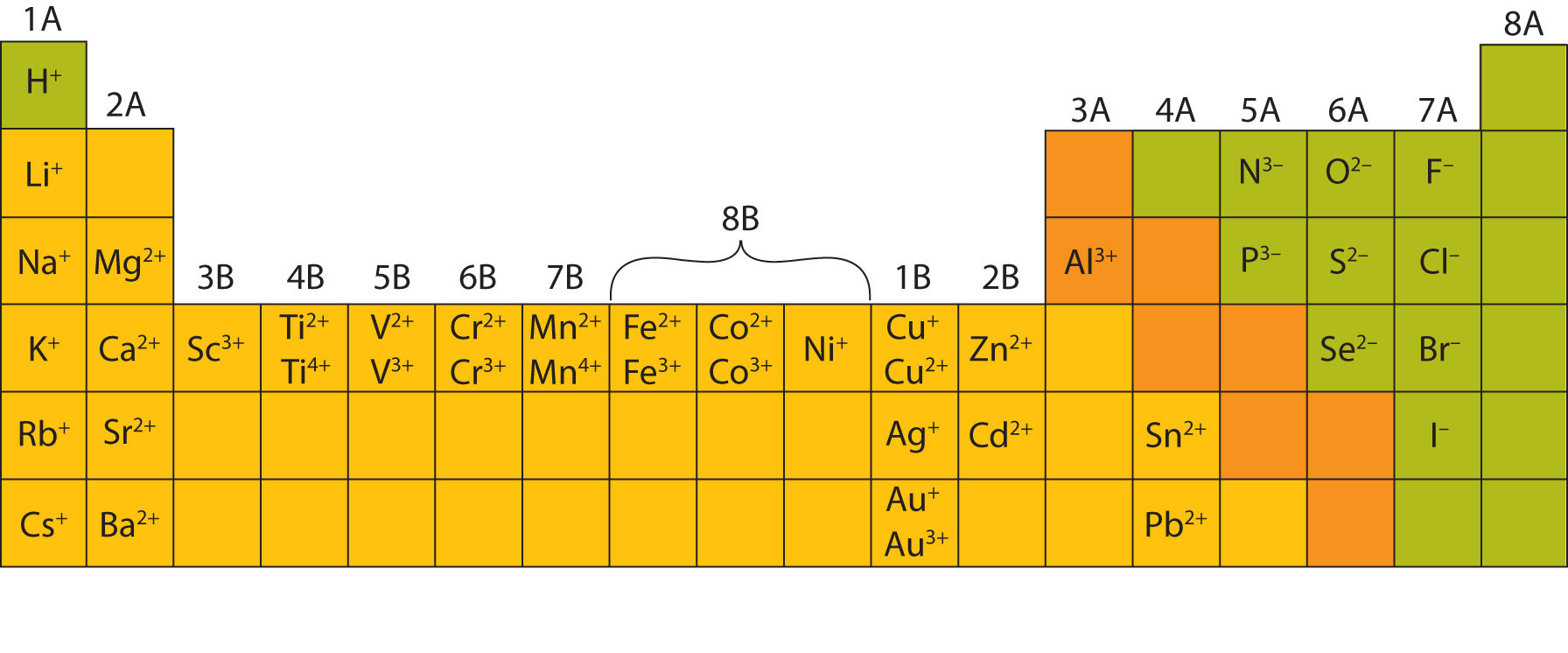
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
